Mastering the Atomic Nucleus: Rutherford Theory, Scattering Graphs, and Solved Problems for NEET/JEE/CBSE
Rutherford's Alpha Scattering Experiment: Complete Theory, Graph Analysis & Practice MCQs
Section 1: The Failure of the Thomson Model & The Alpha Particle
Theory & Concept
Before Rutherford, the Thomson Model (Plum Pudding Model) suggested that positive charge was spread uniformly throughout the atom like a “pudding,” with electrons embedded like “plums.” However, this model could not explain why atoms emit specific spectral lines or why high-energy particles would bounce back from a “soft” diffuse charge.
To probe the atom, Rutherford used Alpha Particles. An Alpha particle is essentially a Helium nucleus. When a Helium atom loses both its electrons, it becomes a bare nucleus with a significant positive charge and mass.
Section 2 Rutherford Scattering and the Atomic Nucleus
a. Introduction to Rutherford Scattering
The Rutherford Scattering Experiment is the foundational study that unveiled the internal structure of the atom. By observing how alpha particles (positively charged helium nuclei) deflected when hitting a thin gold foil, scientists discovered the existence of a dense, positively charged center called the nucleus.
Core Concepts:
Alpha Particle Kinetic Energy: The energy of the moving particle before it interacts with the nucleus.
Electrostatic Potential Energy: The energy of repulsion created between the positive alpha particle and the positive nucleus.
Distance of Closest Approach: The minimum distance an alpha particle reaches before the repulsive force stops its forward motion and pushes it back.
b. Estimating Nuclear Size (Initial Approximation)
In classical physics models, the size of the nucleus was first estimated by assuming the alpha particle’s total energy is converted entirely into potential energy at the moment it stops.
The Energy Equivalence Principle:
To find the distance of closest approach (d), we equate the initial kinetic energy to the potential energy at that point:
Kinetic Energy (K.E.) = Potential Energy (P.E.) (1/2) * m * v² = (k * q1 * q2) / d
Where:
m = mass of the alpha particle
v = velocity of the alpha particle
k = Coulomb’s constant
q1, q2 = charges of the alpha particle and the nucleus
d = nearest distance of closest approach
Initially, researchers believed this distance (d) was roughly equal to the radius of the nucleus.
c. Limitations and Refined Understanding
Further analysis showed that the classical calculation provided only a rough approximation. Accurate experimental data later revealed a significant discrepancy:
The 10x Rule: The actual “nearest distance of closest approach” is approximately ten times larger than the actual physical size of the nucleus.
Why it matters: This indicates that alpha particles do not actually “touch” the nucleus during these scattering events; they are repelled by the strong electrostatic field long before physical contact occurs.
Complexity: Particle-nucleus interactions are more complex than simple energy equivalence and involve trajectories that require advanced modeling.
d. Scientific Impact and Evolution of Atomic Models
Despite the initial numerical inaccuracies regarding size, the experiment was a monumental success:
Model Shift: It successfully disproved the “Plum Pudding” model and established the Nuclear Model of the atom.
Provisional Science: It serves as a classic example of how early approximations act as “stepping stones” for deeper investigations.
Modern Perspective: Today, nuclear physics uses refined scattering interpretations and quantum mechanics to measure nuclear dimensions with extreme precision.
Definitions
Alpha Particle (𝜶): A subatomic particle consisting of 2 protons and 2 neutrons. It is represented as He²⁺.
Charge of 𝜶-particle: q = +2e = +3.2 × 10⁻¹⁹ C.
Mass of 𝜶-particle: approximately 6.64 × 10⁻²⁷ kg (4 times the mass of a hydrogen proton).
Practice MCQ
Q1. Why did Rutherford choose Alpha particles for his scattering experiment?
A) Because they are neutral and won’t be repelled.
B) Because they have high mass and high kinetic energy to penetrate the atom.
C) Because they are negatively charged and attracted to the nucleus.
D) Because they are the smallest particles known.
Answer: B. Detailed Explanation: Alpha particles are heavy (compared to electrons) and possess high velocity (kinetic energy), allowing them to come very close to the nucleus to “feel” its repulsive force without being easily deflected by light electrons.
Section 3: The Gold Foil Experiment Setup & Observations
Theory & Concept
Rutherford directed a beam of 5 MeV Alpha particles at a thin Gold Foil (about 2.1 × 10⁻⁷ m thick). Gold was chosen because it is highly malleable (can be made extremely thin) and has a high atomic number (Z = 79), providing a strong positive charge to repel the Alpha particles.
Key Observations
99.86% of particles passed straight through without any deflection.
About 0.14% deflected by more than 1°.
About 1 in 8000 particles deflected by more than 90° (back-scattering).
Conclusions
Empty Space: Since most particles passed through, the atom is mostly empty space.
The Nucleus: Since some heavy particles bounced back, there must be a tiny, dense, positively charged region at the center called the Nucleus.
Practice MCQ
Q2. What observation led Rutherford to conclude that the nucleus is positively charged?
A) Most particles passed straight through.
B) Particles were attracted and stuck to the foil.
C) Some particles were deflected at large angles or bounced back.
D) Electrons were knocked out of the gold foil.
Answer: C. Detailed Explanation: Since Alpha particles are positive (+2e), they can only be turned back by a very strong repulsive force. Like charges repel, so the central mass must be positive.
Section 4: Impact Parameter & Scattering Angle
Theory & Concept
The trajectory of an alpha particle depends on how “centered” it is towards the nucleus. This is quantified by the Impact Parameter.
Definitions
Impact Parameter (b): The perpendicular distance of the initial velocity vector of the alpha particle from the center of the nucleus.
Scattering Angle (θ): The angle by which the alpha particle is deflected from its original path.
Formulas
The relationship between the Impact Parameter (b) and Scattering Angle (θ) is:
b = [1 / (4𝜋𝜀₀)] × [Z e² cot(θ/2) / K]
Where:
Z = Atomic number of foil.
e = Electronic charge.
K = Kinetic energy of the alpha particle.
cot(θ/2) = Trigonometric cotangent of half the scattering angle.
Key Logic
If b = 0 (Head-on collision), then θ = 180° (Particle bounces straight back).
If b is large, then θ ≈ 0° (Particle passes straight through).
Practice MCQ
Q3. If the impact parameter (b) is increased, what happens to the scattering angle (θ)?
A) θ increases.
B) θ decreases.
C) θ remains constant.
D) θ becomes 180°.
Answer: B. Detailed Explanation: A larger impact parameter means the particle is further from the nucleus. The repulsive force is weaker at greater distances, resulting in less deflection (smaller angle).
Section 5: Distance of Closest Approach (Size of Nucleus)
Theory & Concept
In a head-on collision (b=0), the alpha particle moves directly toward the nucleus. As it gets closer, it slows down because of electrostatic repulsion. At a certain distance, its kinetic energy becomes zero and is entirely converted into electrostatic potential energy. It then turns back.
Formulas
Using the Principle of Conservation of Energy:
Initial Kinetic Energy (K) = Electrostatic Potential Energy (U)
K = [1 / (4𝜋𝜀₀)] × [(2e)(Ze) / r₀]
Rearranging for the Distance of Closest Approach (r₀):
r₀ = [1 / (4𝜋𝜀₀)] × [2Ze² / K]
Where:
r₀ = Distance of closest approach.
1 / (4𝜋𝜀₀) ≈ 9 × 10⁹ N·m²/C².
Numerical Insight
For a 5 MeV alpha particle hitting gold (Z=79), r₀ is approximately 4.13 × 10⁻¹⁴ m. This sets an upper limit on the size of the nucleus.
Practice MCQ
Q4. At the distance of closest approach, the velocity of the alpha particle is:
A) Maximum.
B) Half of the initial velocity.
C) Zero.
D) Equal to the speed of light.
Answer: C. Detailed Explanation: To reverse direction, the particle must momentarily come to a complete stop. At this point, all Kinetic Energy is converted to Potential Energy.
Section 6: Rutherford’s Atomic Model Summary
Key Features
Central Nucleus: All positive charge and nearly 99.9% of the mass is concentrated in the nucleus.
Size: The size of the nucleus (~10⁻¹⁵ m) is very small compared to the size of the atom (~10⁻¹⁰ m).
Orbiting Electrons: Electrons revolve around the nucleus in circular orbits, much like planets around the sun.
Electrostatic Attraction: The centripetal force required for the electrons to orbit is provided by the electrostatic attraction between the positive nucleus and negative electrons.
Definitions
Femtometer (fm): Also called a Fermi. 1 fm = 10⁻¹⁵ m. This is the standard unit for nuclear distances.
Angstrom (Å): 1 Å = 10⁻¹⁰ m. This is the standard unit for atomic distances.
Practice MCQ
Q5. According to Rutherford’s model, what prevents the electrons from flying away from the nucleus?
A) Gravitational force.
B) Magnetic force.
C) Electrostatic force of attraction.
D) Nuclear force.
Answer: C. Detailed Explanation: The positive nucleus pulls the negative electrons inward. This force acts as the centripetal force that keeps the electron in its circular path.
Section 7: Scattering vs. Collision (Mechanical Analogy)
Theory & Concept
Rutherford scattering is often compared to Billiard Ball collisions. However, there is a major difference:
Mechanical Collision: Requires physical contact between surfaces.
Rutherford Scattering: Does not require contact. The particles interact through an “action-at-a-distance” force (Electrostatic force).
Practice MCQ
Q6. Which law of physics governs the path of the alpha particle as it nears the nucleus?
A) Newton’s Law of Cooling.
B) Ohm’s Law.
C) Coulomb’s Law of Electrostatics.
D) Faraday’s Law.
Answer: C. Detailed Explanation: Coulomb’s Law (F = k q₁q₂ / r²) describes the repulsion between the positive alpha particle and the positive gold nucleus.
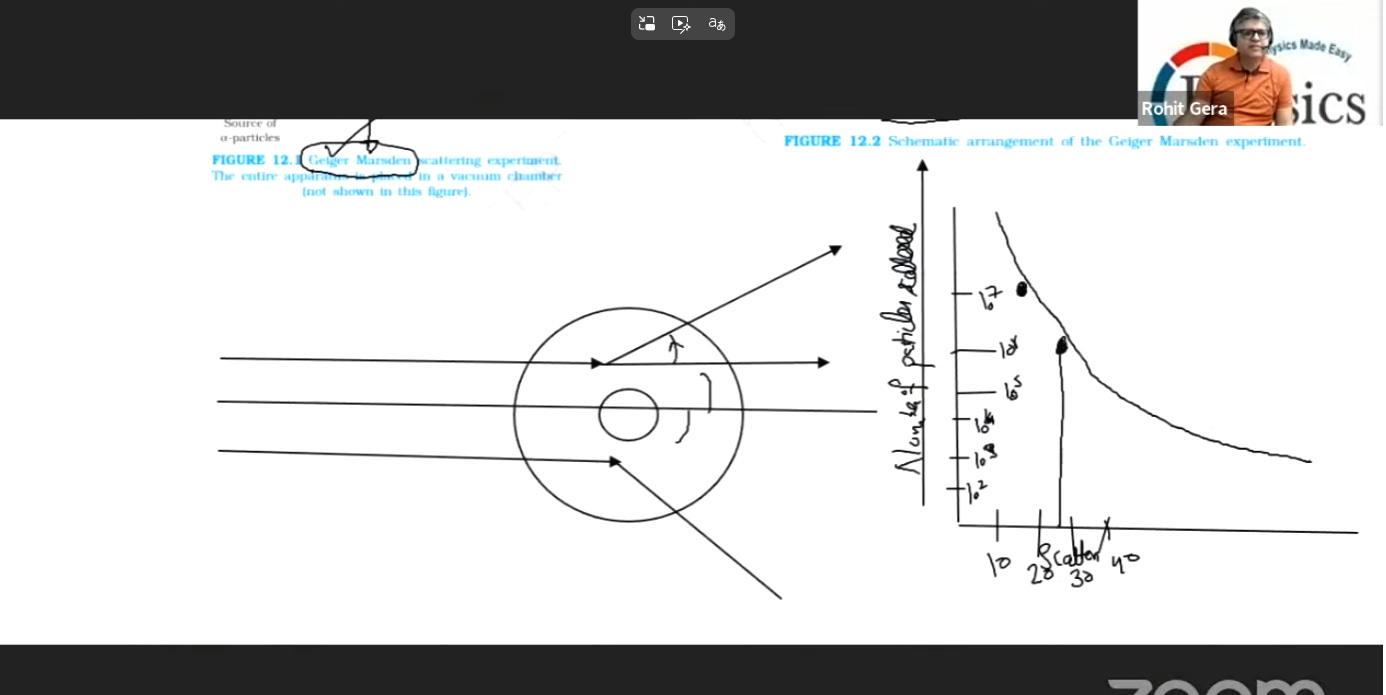
Section 8 : GRAPH OF NUMBER OF PARTICLES SCATTERED VERSUS SCATERRING ANGLE
This graph represents the experimental data from the Geiger-Marsden experiment, which provided the empirical evidence for Rutherford’s nuclear model of the atom.
1. Understanding the Graph Axes
Y-Axis (Number of Particles Scattered): This represents the quantity of alpha particles detected at a specific angle. It is plotted on a logarithmic scale (10², 10³, 10⁴, etc.) to accommodate the massive variation in data points—from millions of particles to just a few hundred.
X-Axis (Scattering Angle, θ): This measures the angle of deflection in degrees (°) from the original straight path.
2. Key Observations and Data Interpretation
Dominance of Small Angles: The graph is highest at the far left (near 10°–20°), showing millions of particles (10⁶ to 10⁷). This indicates that the vast majority of alpha particles pass through the gold foil with almost no deflection.
Rapid Decline: As the scattering angle (θ) increases, the number of particles (N) drops drastically. By 40°, the count falls to the hundreds (10²).
Rare Large-Angle Scattering: Although not shown to the full extent of the x-axis in this specific snippet, the data confirms that very few particles (about 1 in 8,000) deflect at angles greater than 90° or bounce back.
3. Physical Conclusions and Implications
Rutherford used this specific distribution of data to establish the following principles:
Atomic Void: Since most particles pass straight through (high N at small θ), the atom must consist mostly of empty space.
The Nucleus: Large-angle deflections can only occur if the alpha particle encounters a tiny, extremely dense, and positively charged center. If the charge were spread out (as in the Thomson model), such sharp deflections would be impossible.
Coulombic Interaction: The scattering is governed by electrostatic repulsion (Coulomb’s Law) between the +2e charge of the alpha particle and the +Ze charge of the gold nucleus.
4. Mathematical Relationship
The experimental curve follows a specific mathematical law derived from classical mechanics:
N(θ) ∝ 1 / sin⁴(θ/2)
N(θ): Number of particles scattered at angle θ.
sin⁴(θ/2): The fourth power of the sine of half the scattering angle.
This inverse relationship explains why the graph plummet so sharply; as the angle increases even slightly, the denominator grows significantly, causing the number of detected particles to vanish.
Practice MCQ: Mathematical Relationship in Alpha Scattering
Question: In the Rutherford Alpha Particle Scattering experiment, the number of particles N(θ) scattered at a specific angle θ follows a very specific mathematical law. Which of the following correctly represents the relationship between N(θ) and the scattering angle θ?
A) N(θ) ∝ sin⁴(θ/2)
B) N(θ) ∝ 1 / [sin(θ/2)]
C) N(θ) ∝ 1 / [sin⁴(θ/2)]
D) N(θ) ∝ cos²(θ)
Correct Answer: C
Detailed Explanation :
The Relationship: The experiment shows that the number of scattered particles (N) is inversely proportional to the fourth power of the sine of half the scattering angle.
The Formula: N(θ) ∝ 1 / [sin⁴(θ/2)]
Why Option C is Correct: This “Inverse Fourth Power” law explains why the graph drops so steeply. When the angle θ is small, sin(θ/2) is very small, making N(θ) extremely large (millions of particles). As the angle increases, the denominator grows rapidly, causing the number of detected particles to plummet to near zero at large angles.
Scientific Basis: This relationship is derived from Coulomb’s Law of electrostatics, which governs the repulsion between the positive alpha particle and the positive gold nucleus.
Section 8: The Geiger-Marsden Experiment & Electrostatic Energy Conservation
Section a: Overview of the Geiger-Marsden Experiment
Theory & Significance
The Geiger-Marsden experiment is a cornerstone of nuclear physics that builds upon the Rutherford scattering model. It investigates how Alpha particles interact with the heavy, positively charged nuclei of atoms (usually gold). This experiment is vital because it proves that the electrostatic field is a conservative field, meaning total mechanical energy (Kinetic + Potential) remains constant throughout the particle’s journey.
Definitions
Alpha Particle (𝜶): A helium nucleus carrying a +2e positive charge.
Heavy Nucleus: The target (e.g., Gold) carrying a positive charge of +Ze.
Electrostatic Repulsion: The force that pushes the positive alpha particle away from the positive nucleus, preventing a direct collision.
Section b: The Dynamics of Particle Interaction
Concept of Approach
When an alpha particle is fired at a nucleus, it possesses high Kinetic Energy (KE). As it nears the nucleus, the Coulomb interaction (repulsion) slows it down.
The particle travels toward the nucleus but never touches it.
It reaches a minimum distance, called the Distance of Closest Approach (d), where its velocity becomes zero.
At distance d, the particle’s KE is 100% converted into Electrostatic Potential Energy (PE).
The particle then reverses its direction, accelerating away as the potential energy converts back into kinetic energy.
Key Points
Repulsive Coulomb Force: This force governs the entire scattering path.
Scattering Angle: The angle at which the particle deviates depends on its initial energy and the charge of the nucleus.
Section c: Mathematical Relationships & Energy Conservation
The Energy Balance Equation
In a conservative field, the initial energy equals the energy at the point of stopping. This is expressed using the following Unicode-friendly formula:
KE = PE = [1 / (4𝜋𝜀₀)] × [ (Z × 2e) / d ]
Variable Breakdown
Z: Atomic number of the target nucleus.
2e: Charge of the alpha particle.
d: Minimum distance of approach.
𝜀₀: Permittivity of free space.
Mathematical Insight
Inverse Proportionality: The distance d is inversely proportional to the Kinetic Energy (KE).
Logic: Higher KE ➔ Faster particle ➔ Gets closer to the nucleus ➔ Smaller d.
Section d: Practice MCQs (Test Your Knowledge)
Q1. What happens to the Kinetic Energy (KE) of an alpha particle at the distance of closest approach (d)?
A) It reaches its maximum value.
B) It is completely converted into Electrostatic Potential Energy.
C) It is lost as heat to the gold foil.
D) It remains constant throughout the interaction.
Answer: B. Explanation: Because the electrostatic field is conservative, KE is transformed into PE at the stopping point (d) without any energy loss.
Q2. If you double the initial Kinetic Energy of the alpha particle, what happens to the distance of closest approach (d)?
A) d doubles.
B) d stays the same.
C) d is reduced to half.
D) d becomes zero.
Answer: C. Explanation: Since d is inversely proportional to KE, increasing the energy causes the particle to penetrate deeper (closer) to the nucleus, thus reducing the distance d.
Section 5: Summary for Revision
Alpha Particle Charge: +2e.
Nucleus Charge: +Ze.
Energy Conversion: KE ➔ PE (at distance d) ➔ KE (after scattering).
Field Type: Conservative Electrostatic Field.
Key Law: Conservation of Total Mechanical Energy.
📚 Advanced Physics MCQ: Assertion & Reasoning
Instructions: In the following question, a statement of Assertion (A) is followed by a statement of Reason (R). Choose the correct option that describes the relationship between them.
Assertion (A): In the Rutherford scattering experiment, the observation that a very small fraction (approximately 1 in 12,000) of alpha particles rebounded back at large angles confirmed that the positive charge of an atom is concentrated in an extremely small volume called the nucleus.
Reason (R): According to the Plum Pudding Model, alpha particles should have rebounded even more frequently because the positive charge was uniformly spread throughout the entire volume of the atom, creating a dense barrier.
Select the Correct Option:
A) Both (A) and (R) are true, and (R) is the correct explanation of (A).
B) Both (A) and (R) are true, but (R) is NOT the correct explanation of (A).
C) (A) is true, but (R) is false.
D) (A) is false, but (R) is true.
✅ Correct Answer: C
Detailed Analytical Explanation :
Assertion (A) is TRUE: The rarity of the rebound (backward scattering) was the primary evidence used to prove that the nucleus is tiny and dense. If the positive charge were large, more particles would have bounced back.
Reason (R) is FALSE: This is a common misconception. Under the Plum Pudding Model, the positive charge was so diffuse (spread out) that it was expected to be too “soft” to stop or deflect heavy, fast-moving alpha particles. Rutherford actually expected most particles to pass through with minor deflections. He was shocked by the rebounds because the Plum Pudding Model predicted no large-angle scattering at all.