Physics Notes: The Bohr Model & Atomic Spectra
Exploring Hydrogen and Hydrogen-like Systems: Rydberg Calculations, Ionization
Live Class for Recorded The Bohr Model & Atomic Spectra
Physics Notes: The Bohr Model & Atomic Spectra
1. Introduction to the Bohr Model
The Bohr model of the atom introduced the revolutionary idea of quantized electron orbits. This means electrons can only exist in specific, discrete energy levels around the nucleus.
Energy of an electron in the nth orbit:
Eₙ = -13.6 / n² eV
Where n is the principal quantum number (n = 1, 2, 3, ...).
This is one of the most important concepts in the history of physics, marking the transition from classical mechanics to quantum mechanics.
Here is an explanation of the meaning of quantized electron orbits and their immense significance in understanding the atom.
The Meaning of “Quantized Electron Orbits”
Before Niels Bohr proposed his model in 1913, the prevailing view (the Rutherford model) resembled a miniature solar system: negatively charged electrons orbiting a dense, positively charged nucleus, much like planets orbit the sun.
In classical physics, a planet can theoretically orbit the sun at any distance, provided it has the correct speed. Consequently, it could possess any continuous amount of energy.
Bohr broke this rule.
Quantization means “restricted to discrete values.” When we say electron orbits are quantized, it means the electron is forbidden from occupying just any space around the nucleus. Instead, it is restricted to traveling only in certain specifically allowed circular paths (orbits) at fixed distances from the nucleus.
The “Ladder” Analogy
The best way to visualize this is to imagine a ladder:
You can stand on the first rung (n=1), the second rung (n=2), or the third rung (n=3).
You cannot stand in the space between the rungs.
In the Bohr atom:
The rungs of the ladder are the allowed orbits (energy levels).
The variable n (the principal quantum number) is simply the number of the rung.
Because the electron must be on a specific rung, it must possess a specific amount of energy.
Connecting to the Formula
Look at the formula you provided:
En= (−13.6/ n^2) eV
Here, n must not be a positive integer (1, 2, 3...).
If n=1, energy is exactly -13.6 eV.
If n=2, energy is exactly -3.4 eV.
An electron in a hydrogen atom cannot have an energy of -10 eV or -5 eV, because there is no whole number n that produces those values.
In summary, the meaning of quantized orbits is that the electron is locked into specific energy states and cannot exist “in between” them.
2. The Significance of Quantized Orbits
Bohr didn’t just invent this idea because it sounded neat; it was necessary to solve two major crises in physics that classical theories couldn’t explain.
Significance A: Explaining Atomic Stability (Why atoms don’t collapse)
This was the biggest problem with the previous Rutherford model.
According to classical electromagnetic theory, any charged particle that accelerates (moves in a curved path like a circle) must radiate energy in the form of light.In classical physics, an object moving in a circle is constantly changing direction, which means it is perpetually accelerating (centripetal acceleration), even at a constant speed. Maxwell's electromagnetic theory dictates that whenever a charged particle, like an electron, accelerates, it disrupts the electromagnetic field around it, generating outward-propagating ripples. These ripples are electromagnetic waves—essentially light—that carry energy away from the source. Therefore, a classically orbiting electron should continuously radiate away its kinetic energy and rapidly spiral into the nucleus.
If an orbiting electron were constantly radiating energy, it would constantly lose speed. As it lost speed, its orbit would shrink. Within a fraction of a second, the electron would spiral inwardly and crash into the nucleus. Matter as we know it shouldn’t exist.This phrase is not a dramatic exaggeration; it is a literal interpretation of what physics predicted before quantum mechanics. If the classical “death spiral” of the electron were true, the universe would be radically different in three fundamental ways:
No Volume or Structure: An atom is 99.999999999% empty space, defined entirely by the distance at which electrons orbit the nucleus. If electrons crashed into the nucleus, that empty space would vanish. All the matter in the Empire State Building would collapse into a speck of dust, immensely heavy but microscopic. We, planets, and stars could not have physical size or form.
No Chemistry or Molecules: “Matter as we know it” relies on chemical bonding (like H₂O or DNA). Chemistry is essentially the interaction of the outermost electrons of neighboring atoms. If electrons are crashing into their own nuclei, they cannot form stable shells to interact with other atoms. There would be no water, no air, no rocks, and certainly no life.
Instant Instability: The calculated time for this collapse is roughly 10−11 seconds. A universe governed by these classical rules would not last long enough for stars to form, let alone biological evolution. It would instantly collapse into a dense, inert soup of neutrons (formed when electrons smash into protons).
Bohr’s Solution: By quantizing the orbits, Bohr introduced a radical new rule: As long as an electron stays in one of these allowed, quantized orbits (called “stationary states”), it does not radiate energy.
It only radiates or absorbs energy when it jumps from one allowed orbit to another. This arbitrary rule saved the atom from collapsing and explained the stability of matter.
Significance B: Explaining Atomic Spectra (The “Fingerprints” of Elements)
When you heat up a gas (like hydrogen in a neon tube) and pass the light through a prism, you don’t see a continuous rainbow. You see sharp, distinct lines of specific colors separated by darkness. For decades, no one knew why hydrogen always emitted the exact same colors.
Bohr’s Solution: Quantized orbits perfectly explained this.
Electrons usually sit in the lowest energy rung (n=1, the ground state).
When heated, the electron absorbs energy and jumps to a higher rung (e.g., n=3).
Nature prefers the lowest energy state, so the electron immediately falls back down to a lower rung (e.g., from n=3 down to n=2).
Because the rungs are at fixed energy positions, the jump down releases a very specific, fixed amount of energy as a photon of light.
A specific amount of energy corresponds exactly to a specific color of light. Because the orbits (”rungs”) are quantized, the jumps are quantized, and therefore the colors of light emitted are quantized (specific spectral lines).
Significance C: The Gateway to Modern Quantum Mechanics
While the Bohr model turned out to be too simple to work perfectly for atoms larger than hydrogen, the concept of quantization was revolutionary. It was the crucial first step that proved classical physics did not apply at the atomic scale, paving the way for the more complete quantum mechanics of Schrödinger and Heisenberg that followed.
3. Quantum Transitions & Emission Spectra
A fundamental concept in the Bohr model is that electrons can transition between these discrete energy levels.
Transition Rules:
Absorption: An electron absorbs a photon and jumps to a higher energy level.
Emission: An electron drops to a lower energy level and emits a photon.
Energy Difference & Photon Properties:
The energy difference between the initial (n₁) and final (n₂) orbits determines the energy, frequency (ν), and wavelength (λ) of the absorbed or emitted photon.
ΔE = Eₙ₂ - Eₙ₁ = -13.6 (1/n₂² - 1/n₁²) eV
E = hν = hc / λ (Where h is Planck’s constant and c is the speed of light)
Emission Spectrum: The collection of all discrete frequencies (colors) of light emitted by an atom due to these electron transitions forms its unique emission spectrum.
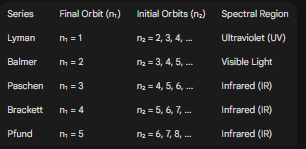
4. Spectral Series of Hydrogen
The emission spectrum of hydrogen consists of several distinct series of spectral lines, classified by the final energy level (n₁) to which the electron falls.
Rydberg Formula: Wavelengths of these lines can be calculated using:
1/λ = R_H (1/n₁² - 1/n₂²)
Where R_H is the Rydberg constant (approximately 1.097 x 10⁷ m⁻¹).
5. Energy Levels & Convergence
Energy vs. Orbit: Electron energy increases (becomes less negative) as the orbit number (n) increases, approaching zero.
Energy Spacing: The energy difference between adjacent orbits decreases as n increases. This causes spectral lines in a series to crowd together, approaching a series limit at the shortest wavelength.
6. Wavelength Extremes & Spectral Regions
Photon Energy & Wavelength: Higher energy transitions correspond to higher frequency and shorter wavelength photons.
Lyman series (transitions to n=1) involves large energy changes -> UV light.
Balmer series (transitions to n=2) involves smaller energy changes -> Visible light.
Series Limits:
Minimum Wavelength (λₘᵢₙ): Corresponds to the largest energy transition, from n₂ = ∞ to n₁. This is the series limit.
Maximum Wavelength (λₘₐₓ): Corresponds to the smallest energy transition, between adjacent orbits (n₂ = n₁ + 1 to n₁).
7. Ionization & Kinetic Energy
Ionization: When an electron absorbs enough energy to reach the n = ∞ state (E = 0), it becomes free from the atom. The atom is ionized.
Ionization Energy: The minimum energy required to remove an electron from the ground state (n=1). For hydrogen, this is 13.6 eV.
Kinetic Energy: If the absorbed photon has energy greater than the ionization energy, the excess energy becomes the kinetic energy of the ejected electron.
KE = Eₚₕₒₜₒₙ - Eᵢₒₙᵢzₐₜᵢₒₙ
8. Hydrogen-like Ions
The Bohr model can be applied to ions with a single electron (e.g., He⁺, Li²⁺) by accounting for the nuclear charge (Z).
Modified Formulas:
Energy: Eₙ = -13.6 * (Z² / n²) eV
Radius: rₙ = (n² * 0.529 Å) / Z
Velocity: vₙ = (2.19 x 10⁶ m/s * Z) / n
9. Absorption Spectra & Astrophysics
Absorption Spectrum: Produced when atoms absorb specific frequencies of light, causing electrons to jump to higher energy levels. This results in dark lines on a continuous spectrum background.
Astrophysical Importance: Absorption lines in starlight act as “fingerprints,” allowing astronomers to identify the chemical elements present in stars and interstellar gas clouds.
10. Calculating Spectral Lines
The total number of possible spectral lines emitted when an electron is in an excited state n can be calculated using the formula:
Number of lines = n(n - 1) / 2
Here is the section rewritten using Unicode symbols for headings, variables, operators, and emphasis, making it ready for direct copy-pasting.
Below is a detailed example illustrating this concept.
𝐄𝐱𝐚𝐦𝐩𝐥𝐞: 𝐄𝐥𝐞𝐜𝐭𝐫𝐨𝐧 𝐢𝐧 𝐭𝐡𝐞 𝟒𝐭𝐡 𝐄𝐱𝐜𝐢𝐭𝐞𝐝 𝐒𝐭𝐚𝐭𝐞 (𝑛 = 𝟒)
Let’s imagine a sample of hydrogen atoms where the electrons have been excited to the principal quantum number 𝑛 = 𝟒. We want to calculate the total number of different spectral lines that could be observed as these electrons de-excite back down toward the ground state (𝑛 = 𝟏).
𝟏. 𝐔𝐬𝐢𝐧𝐠 𝐭𝐡𝐞 𝐅𝐨𝐫𝐦𝐮𝐥𝐚 (𝐓𝐡𝐞 𝐅𝐚𝐬𝐭 𝐌𝐞𝐭𝐡𝐨𝐝)
We can directly apply the formula for the total number of possible spectral lines (𝑁):
• Given excited state: 𝑛 = 𝟒 • Formula: 𝑁 = 𝑛(𝑛 − 𝟏) / 𝟐 • Substitution: 𝑁 = 𝟒(𝟒 − 𝟏) / 𝟐 • Calculation: 𝑁 = 𝟒 × 𝟑 / 𝟐 = 𝟏𝟐 / 𝟐 = 𝟔
➡️ 𝐑𝐞𝐬𝐮𝐥𝐭: There will be a total of 𝟔 𝐝𝐢𝐬𝐭𝐢𝐧𝐜𝐭 𝐬𝐩𝐞𝐜𝐭𝐫𝐚𝐥 𝐥𝐢𝐧𝐞𝐬 observed.
𝟐. 𝐕𝐢𝐬𝐮𝐚𝐥𝐢𝐳𝐢𝐧𝐠 𝐭𝐡𝐞 𝐓𝐫𝐚𝐧𝐬𝐢𝐭𝐢𝐨𝐧𝐬 (𝐓𝐡𝐞 𝐕𝐞𝐫𝐢𝐟𝐢𝐜𝐚𝐭𝐢𝐨𝐧 𝐌𝐞𝐭𝐡𝐨𝐝)
To understand why the answer is 𝟔, we list every possible distinct downward transition an electron can make starting from 𝑛=𝟒 and eventually landing at 𝑛=𝟏.
Remember, an electron doesn’t have to jump straight to the bottom; it can cascade down step-by-step. Every distinct jump produces a photon of a unique energy (a unique spectral line).
Possible Transitions:
• 𝐒𝐭𝐚𝐫𝐭𝐢𝐧𝐠 𝐟𝐫𝐨𝐦 𝑛 = 𝟒: 𝟏. Transition 𝑛 = 𝟒 → 𝑛 = 𝟑 (Emits a photon in the Paschen series) 𝟐. Transition 𝑛 = 𝟒 → 𝑛 = 𝟐 (Emits a photon in the Balmer series) 𝟑. Transition 𝑛 = 𝟒 → 𝑛 = 𝟏 (Emits a photon in the Lyman series)
• If an electron landed on 𝑛=𝟑, it can fall further: 𝟒. Transition 𝑛 = 𝟑 → 𝑛 = 𝟐 (Emits a photon in the Balmer series) 𝟓. Transition 𝑛 = 𝟑 → 𝑛 = 𝟏 (Emits a photon in the Lyman series)
• If an electron landed on 𝑛=𝟐, it can fall further: 𝟔. Transition 𝑛 = 𝟐 → 𝑛 = 𝟏 (Emits a photon in the Lyman series)
When we count these distinct transitions (𝟒→𝟑, 𝟒→𝟐, 𝟒→𝟏, 𝟑→𝟐, 𝟑→𝟏, 𝟐→𝟏), we find exactly 𝟔 possible lines, confirming the formula works.
Key Takeaways:
The Bohr model’s concept of quantized energy levels is crucial for explaining atomic stability and spectra.
Spectral lines originate from electron transitions between these fixed energy levels.
The energy difference of the transition determines the wavelength of the emitted or absorbed light.
Hydrogen’s spectrum is organized into distinct series (Lyman, Balmer, etc.) based on the final energy level.
Here is the tricky Assertion-Reasoning question rewritten using Unicode symb
𝐑𝐞𝐚𝐬𝐨𝐧𝐢𝐧𝐠 𝐚𝐧𝐝 𝐀𝐬𝐬𝐞𝐫𝐭𝐢𝐨𝐧 𝐐𝐮𝐞𝐬𝐭𝐢𝐨𝐧
𝐃𝐢𝐫𝐞𝐜𝐭𝐢𝐨𝐧𝐬: In the following question, a statement of Assertion (A) is followed by a statement of Reason (R). Choose the correct alternative from the following options:
𝐀) Both Assertion (A) and Reason (R) are true, and Reason (R) is the correct explanation of Assertion (A).
𝐁) Both Assertion (A) and Reason (R) are true, but Reason (R) is NOT the correct explanation of Assertion (A).
𝐂) Assertion (A) is true, but Reason (R) is false.
𝐃) Assertion (A) is false, but Reason (R) is true.
𝐀𝐬𝐬𝐞𝐫𝐭𝐢𝐨𝐧 (𝐀): A sample of hydrogen gas, initially in the ground state, absorbs photons with an energy of 𝟏𝟐.𝟓 eV. Upon subsequent de-excitation, the emission spectrum of this sample will contain spectral lines belonging to the Lyman, Balmer, and Paschen series only.
𝐑𝐞𝐚𝐬𝐨𝐧 (𝐑): According to the Bohr model, the number of possible spectral lines emitted when an electron is in the 𝑛ᵗʰ excited state is given by the formula 𝑛(𝑛−𝟏)/𝟐, and for a transition to occur, the absorbed photon’s energy must exactly match the energy difference between two specific discrete energy levels.
𝐒𝐨𝐥𝐮𝐭𝐢𝐨𝐧 𝐚𝐧𝐝 𝐄𝐱𝐩𝐥𝐚𝐧𝐚𝐭𝐢𝐨𝐧
𝐂𝐨𝐫𝐫𝐞𝐜𝐭 𝐀𝐧𝐬𝐰𝐞𝐫: 𝐃) 𝐀𝐬𝐬𝐞𝐫𝐭𝐢𝐨𝐧 (𝐀) 𝐢𝐬 𝐟𝐚𝐥𝐬𝐞, 𝐛𝐮𝐭 𝐑𝐞𝐚𝐬𝐨𝐧 (𝐑) 𝐢𝐬 𝐭𝐫𝐮𝐞.
𝐃𝐞𝐭𝐚𝐢𝐥𝐞𝐝 𝐄𝐱𝐩𝐥𝐚𝐧𝐚𝐭𝐢𝐨𝐧:
𝟏. 𝐀𝐧𝐚𝐥𝐲𝐳𝐢𝐧𝐠 𝐭𝐡𝐞 𝐑𝐞𝐚𝐬𝐨𝐧 (𝐑): This statement consists of two parts, both drawn directly from physics principles:
“The number of possible spectral lines emitted when an electron is in the 𝑛ᵗʰ excited state is given by the formula 𝑛(𝑛−𝟏)/𝟐.” (This is a correct standard formula).
“...for a transition to occur, the absorbed photon’s energy must exactly match the energy difference between two specific discrete energy levels.” (This is the fundamental quantum rule for absorption). Therefore, the Reason (R) is entirely a true statement.
𝟐. 𝐀𝐧𝐚𝐥𝐲𝐳𝐢𝐧𝐠 𝐭𝐡𝐞 𝐀𝐬𝐬𝐞𝐫𝐭𝐢𝐨𝐧 (𝐀): This is where the trick lies. Let’s break it down step-by-step.
• 𝐒𝐭𝐞𝐩 𝟏: 𝐃𝐞𝐭𝐞𝐫𝐦𝐢𝐧𝐞 𝐭𝐡𝐞 𝐞𝐧𝐞𝐫𝐠𝐲 𝐥𝐞𝐯𝐞𝐥𝐬 𝐨𝐟 𝐇𝐲𝐝𝐫𝐨𝐠𝐞𝐧. Using the formula 𝐸ₙ = −𝟏𝟑.𝟔 eV / 𝑛²:
𝑛=𝟏 (Ground state): 𝐸₁ = −𝟏𝟑.𝟔 eV
𝑛=𝟐: 𝐸₂ = −𝟏𝟑.𝟔 / 𝟒 = −𝟑.𝟒 eV
𝑛=𝟑: 𝐸₃ = −𝟏𝟑.𝟔 / 𝟗 ≈ −𝟏.𝟓𝟏 eV
𝑛=𝟒: 𝐸₄ = −𝟏𝟑.𝟔 / 𝟏𝟔 = −𝟎.𝟖𝟓 eV
• 𝐒𝐭𝐞𝐩 𝟐: 𝐂𝐚𝐥𝐜𝐮𝐥𝐚𝐭𝐞 𝐭𝐡𝐞 𝐞𝐧𝐞𝐫𝐠𝐲 𝐫𝐞𝐪𝐮𝐢𝐫𝐞𝐝 𝐟𝐨𝐫 𝐞𝐱𝐜𝐢𝐭𝐚𝐭𝐢𝐨𝐧 𝐟𝐫𝐨𝐦 𝐭𝐡𝐞 𝐠𝐫𝐨𝐮𝐧𝐝 𝐬𝐭𝐚𝐭𝐞.
To jump from 𝑛=𝟏 to 𝑛=𝟐: Δ𝐸 = 𝐸₂ − 𝐸₁ = −𝟑.𝟒 − (−𝟏𝟑.𝟔) = 𝟏𝟎.𝟐 eV
To jump from 𝑛=𝟏 to 𝑛=𝟑: Δ𝐸 = 𝐸₃ − 𝐸₁ = −𝟏.𝟓𝟏 − (−𝟏𝟑.𝟔) = 𝟏𝟐.𝟎𝟗 eV
To jump from 𝑛=𝟏 to 𝑛=𝟒: Δ𝐸 = 𝐸₄ − 𝐸₁ = −𝟎.𝟖𝟓 − (−𝟏𝟑.𝟔) = 𝟏𝟐.𝟕𝟓 eV
• 𝐒𝐭𝐞𝐩 𝟑: 𝐀𝐧𝐚𝐥𝐲𝐳𝐞 𝐭𝐡𝐞 𝐚𝐛𝐬𝐨𝐫𝐩𝐭𝐢𝐨𝐧. The assertion states the gas is exposed to photons with energy exactly equal to 𝟏𝟐.𝟓 eV. As stated in Reason (R), an electron can only absorb a photon if its energy exactly matches the difference between two levels.
Is 𝟏𝟐.𝟓 eV equal to 𝟏𝟎.𝟐 eV? No.
Is 𝟏𝟐.𝟓 eV equal to 𝟏𝟐.𝟎𝟗 eV? No.
Is 𝟏𝟐.𝟓 eV equal to 𝟏𝟐.𝟕𝟓 eV? No.
Since 𝟏𝟐.𝟓 eV does not match any specific transition energy from the ground state (𝑛=𝟏) to a higher state, the hydrogen atoms will not absorb these photons. The electrons will remain in the ground state.
• 𝐒𝐭𝐞𝐩 𝟒: 𝐀𝐧𝐚𝐥𝐲𝐳𝐞 𝐭𝐡𝐞 𝐞𝐦𝐢𝐬𝐬𝐢𝐨𝐧. Since no absorption occurred, there is no excitation. If there is no excitation, there can be no de-excitation. Therefore, no spectral lines will be emitted.
𝐂𝐨𝐧𝐜𝐥𝐮𝐬𝐢𝐨𝐧 𝐨𝐧 𝐀𝐬𝐬𝐞𝐫𝐭𝐢𝐨𝐧 (𝐀): The assertion claims that spectral lines will be seen in various series. This is false because excitation never happened in the first place due to the energy mismatch.
( Note: Even if the electron had reached 𝑛=𝟑 (requiring 𝟏𝟐.𝟎𝟗 eV), the assertion would still be false because Paschen lines arise from transitions ending at 𝑛=𝟑, which cannot happen if you only start at 𝑛=𝟑.)
Since (A) is False and (R) is True, the correct option is 𝐃.